Types of Reactions:
Combustion: A element or compound will combine with Oxygen to form water and Carbon dioxide.
Double Replacement: Two compounds react to form two new compounds.
Single Replacement: A single element reacts with a compound to form a new element and compound.
Decomposition: A compound is broken down to form two elements.
Combination: Two elements are combined to form a single compound.
Combustion: A element or compound will combine with Oxygen to form water and Carbon dioxide.
Double Replacement: Two compounds react to form two new compounds.
Single Replacement: A single element reacts with a compound to form a new element and compound.
Decomposition: A compound is broken down to form two elements.
Combination: Two elements are combined to form a single compound.
Solubility rules- pg. 344
Activity Series- pg. 333
Activity Series- pg. 333
How to: Net Ionic Equations
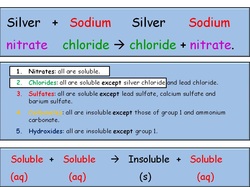
Solubility Rules
Activity Series- Elements can replace anything below them but not above on the list of decreasing re activity
For Halogens the order of decreasing re activity goes down the group
For Halogens the order of decreasing re activity goes down the group
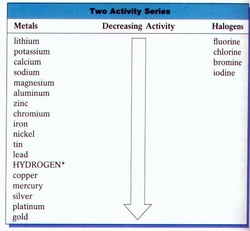
Activity series for metals and halogens
