** q=(m)(deltaT)(Cp)
q:heat
m:mass
deltaT:change in temperature
Cp:specific heat
q:heat
m:mass
deltaT:change in temperature
Cp:specific heat
A substance with a high specific heat will not change temperature easily
Ex: Water
Ex: Water
**Law of Conservation of Energy: States that in any chemical or physical process energy is neither created nor destroyed
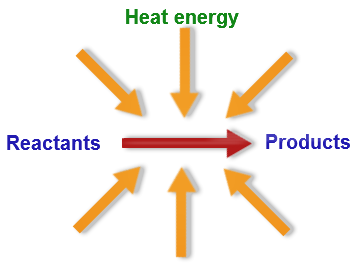
Endothermic vs. Exothermic
Endo-absorbs heat from the surroundings
Exo-releases heat to its surroundings
Endo-absorbs heat from the surroundings
Exo-releases heat to its surroundings
Heat-represented by q, is energy that transfers from one object to another because of a temperature difference between them
*Heat always flows from a warmer object to a cooler object
*Heat always flows from a warmer object to a cooler object
Heat does not always rise! It is transferred, so it flows from warm to cold.
Ex: Your foot stepping on an ice cube, the heat from your foot will flow downward to the ice cube.
Ex: Your foot stepping on an ice cube, the heat from your foot will flow downward to the ice cube.
System-the part of the universe on which you focus your attention
Surroundings-everything else in the universe that is not the system
Surroundings-everything else in the universe that is not the system
1 Calorie = 1000 calories
1 Calorie = 4186 Joules
1 Calorie = 4186 Joules
Heat capacity vs. Specific heat
Heat capacity-the amount of heat needed to increased the temperature of an object exactly 1 degree celsius
Specific heat-the amount of heat it takes to raise the temperature of 1 g of the substance 1 degree celsius
Heat capacity-the amount of heat needed to increased the temperature of an object exactly 1 degree celsius
Specific heat-the amount of heat it takes to raise the temperature of 1 g of the substance 1 degree celsius
Enthalpy-the heat content of a system at a constant pressure
Entropy-a measure of the disorder of a system
Entropy-a measure of the disorder of a system