1 atm=760mmHg=101.3kPa
Evaporation occurs when the particles with the highest kinetic energy on the top layer escape (not boiling)
Boiling point: The temperature at which the vapor pressure of the liquid is just equal to the external pressure on the liquid
Normal Boiling point: The boiling point of a liquid at a pressure of 101.3 kPa or one atmosphere (atm)
Melting point: The temperature when a solid turns into a liquid
Boiling point: The temperature at which the vapor pressure of the liquid is just equal to the external pressure on the liquid
Normal Boiling point: The boiling point of a liquid at a pressure of 101.3 kPa or one atmosphere (atm)
Melting point: The temperature when a solid turns into a liquid
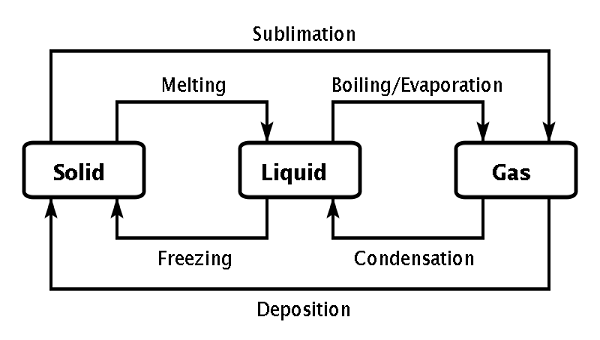
Chapter 13 Concept Map
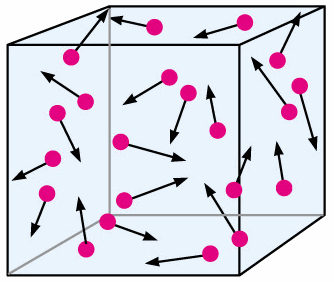
According to kinetic theory: All particles in any state are in constant motion
Kinetic Energy: The energy a substance has based on its motion
The Kelvin temperature scale and average kinetic energy are directly proportional
0 K (absolute zero) is when particles theoretically stop moving
0 K (absolute zero) is when particles theoretically stop moving
Vapor Pressure: The measure of the force exerted by a gas above a liquid
Measured with: Manometer
Atmospheric Pressure: The pressure that results from the collisions of atoms and molecules in air with objects
Measure with: Barometer
Measured with: Manometer
Atmospheric Pressure: The pressure that results from the collisions of atoms and molecules in air with objects
Measure with: Barometer
A gas' particles move randomly and their collisions are perfectly elastic
A gas has no definite shape of volume
A gas has the ability to flow so it is a fluid
A gas has no definite shape of volume
A gas has the ability to flow so it is a fluid
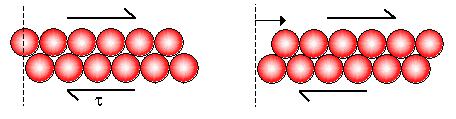
A liquid's particles slip n' slide past eachother
A liquid has a definite volume but no definite shape
A liquid has the ability to flow so it is a fluid
A liquid has a definite volume but no definite shape
A liquid has the ability to flow so it is a fluid
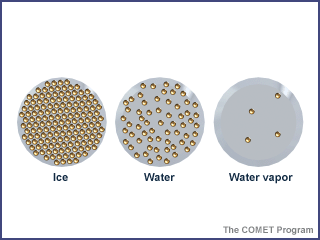
A solid's particles vibrate in a pack
A solid has a definite volume and shape
A solid has a definite volume and shape