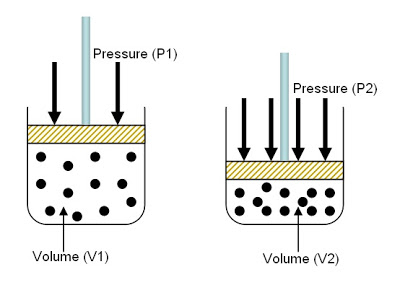
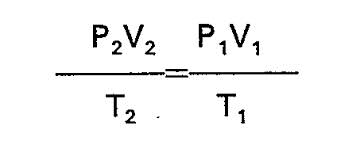There are three main gas laws: Boyle's, Charle's, and GL's
To change celsius to Kelvin add 237.
Pressure should be in kPa's but it can be in atms
Volume should be in Liters
1) List everything you know and put a question mark next to what you are solving for.
2) When finding for a variable, circle it and use the arrow method to get it alone.
3) Cross out any variables you don't use.
4) Make sure temperature in always in Kelvin.
5) Finally put the numbers in and solve!
Pressure should be in kPa's but it can be in atms
Volume should be in Liters
1) List everything you know and put a question mark next to what you are solving for.
2) When finding for a variable, circle it and use the arrow method to get it alone.
3) Cross out any variables you don't use.
4) Make sure temperature in always in Kelvin.
5) Finally put the numbers in and solve!
Here are some examples of everyday instances that Boyle's law can be seen:
syringe, suction cup, popping a straw, blowing up a basketball, bubble wrap, air bags, IV bags, scuba diving, breathing, popping the top off a water bottle, and plunger
syringe, suction cup, popping a straw, blowing up a basketball, bubble wrap, air bags, IV bags, scuba diving, breathing, popping the top off a water bottle, and plunger
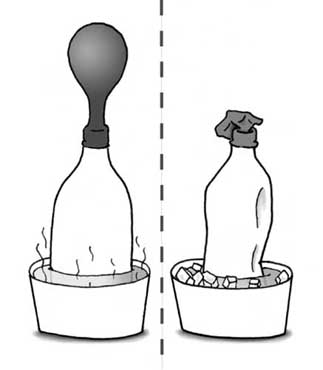
Here are some examples of everyday instances where Charles' law can be seen: blowing up a balloon, taking a balloon in the cold, baking bread, roasting a marshmallow, whistling teapot, hot air balloons, and deflating basketballs in the cold
Here are some examples of everyday instances where GL's law can be seen: silly string cans, CO2 bike cartridges, taking a drill to a lighter, popcorn, torch, aerosol can in fire, whistling teapot, and diesel engines